By Colin Young December 4, 2025
The transition to the revised EU GMP Annex 1 (2022) demands a shift from simply building a clean space to engineering a comprehensive Contamination Control Strategy (CCS). Validation issues are often rooted in the design phase, where planning assumptions misalign with operational reality.
If your facility design does not thoroughly address risks associated with operator interventions or material and equipment transfer to sterile areas, you are potentially compromising your sterility assurance.
Five common shortcomings in cleanroom design
These common design challenges can lead to costly mitigation, arising from regulatory exposure and non-conformance:
Dynamic Pressure Differential Breakdown
The typical ≥10 Pa pressure cascade is a functional dynamic requirement, not a static target If the HVAC system lacks the ability to quickly recover from pressure decay during transient events, the barrier is momentarily compromised. Full compliance demands fast-acting pressure controls. [Annex 1, Section 2.3]
Compromised Aseptic Barrier Integrity
Isolators and RABS systems are the primary sterile barrier, but complexity and frequency of operator interventions are often underestimated. Optimizing design to minimize required interventions proactively reduces contamination risk during Aseptic Process Simulations (APS) and Media Fills. [Annex 1, Section 9.2]
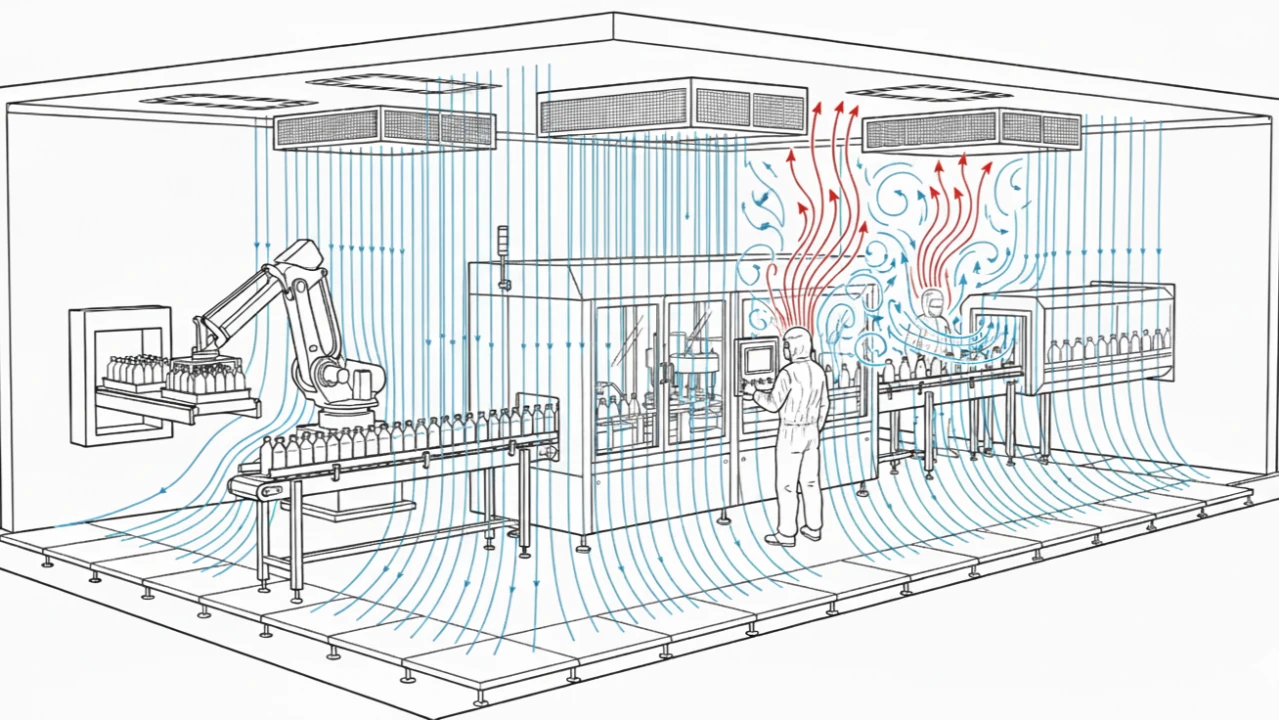
Inadequate Mitigation of Thermal Plumes:
Unidirectional airflow is the cornerstone of Grade A protection. Operators are equivalent to 100W mobile heat sources (Yes, even the ones making slow and deliberate movements!). This thermal load creates buoyant thermal plumes that can actively disrupt unidirectional flow, which could induce turbulence over the critical filling path. This risk should be identified proactively. [Annex 1, Section 6.2.1]
Substandard Surface Efficacy for Vaporized Hydrogen Peroxide (VHP)
Effective VHP decontamination is dependent upon maximum surface contact. Rough finishes on critical stainless-steel surfaces (e.g., Ra>0.3μm) introduce the risk of micro-crevices, creating microbial hideouts perfectly shielded from VHP vapour. [Annex 1, Section 2.1]
Inconsistent Integration of Real-Time Data Integrity
Cleanroom designs often treat the Environmental Monitoring System (EMS) and Building Management System (BMS) as separate data silos. Data integrity and continuous monitoring are explicitly mandated in Annex 1. Inability to design an integrated, secure, 21 CFR Part 11 -compliant data backbone could lead to significant audit findings. [Annex 1, Section 4.3 & 9.3]
The Solution
To ensure qualification success and ongoing operational and sterility assurance, consider mandating the following six engineering requirements as part of the design:
Dynamic Computational Fluid Dynamic (CFD) Analysis
Use CFD modeling to simulate the dynamic worst-case scenarios, including thermal load and simulated movement of operators, confirming the unidirectional flow velocity remains within the qualified range. [Annex 1, Section 6.2]
Enforce Closed-Loop Transfers
Eliminate all open interfaces. Specify No-Touch-Transfer (NTT) for inbound components and an integrated, dynamically controlled Tunnel Conveyor System for outbound sealed product. [Annex 1, Section 4.1 & 4.2]
Design for Minimum Interventions (APS Focus)
Optimize design to reduce the frequency, complexity, and duration of required operator interventions. All critical interventions must be assessed and tested in advance to ensure success during Media Fill. [Annex 1, Section 9.2]
Specify Surface Roughness of <0.3μm
Require all stainless steel (316L) in the Grade A environment to achieve an Ra value of <0.3μm with required 20mm radius internal corners to ensure VHP penetration and cleanability. [Annex 1, Section 2.1]
Qualify Maximum Load Pattern
Ensure the VHP qualification protocol addresses the Worst-Case Load Pattern (maximum tool and component density) to prove the required sterility assurance levels are met. [Annex 1, Section 3.2.2]
Implement Integrated Data & Monitoring Architecture
Mandate a single, secure, 21 CFR Part 11 -compliant data platform that integrates the EMS and BMS/HVAC controls. This ensures all critical physical and microbiological data are protected. [Annex 1, Section 4.3 & 9.3]
Which of these 6 solutions gave your last validation project the most ROI? Let us know!
