The Bio-Based Evolution in Biopharma
There is a common misconception that “bio-based” means “compostable” or “weak.” In the high-stakes world of biopharmaceutical manufacturing, that couldn’t be further from the truth.
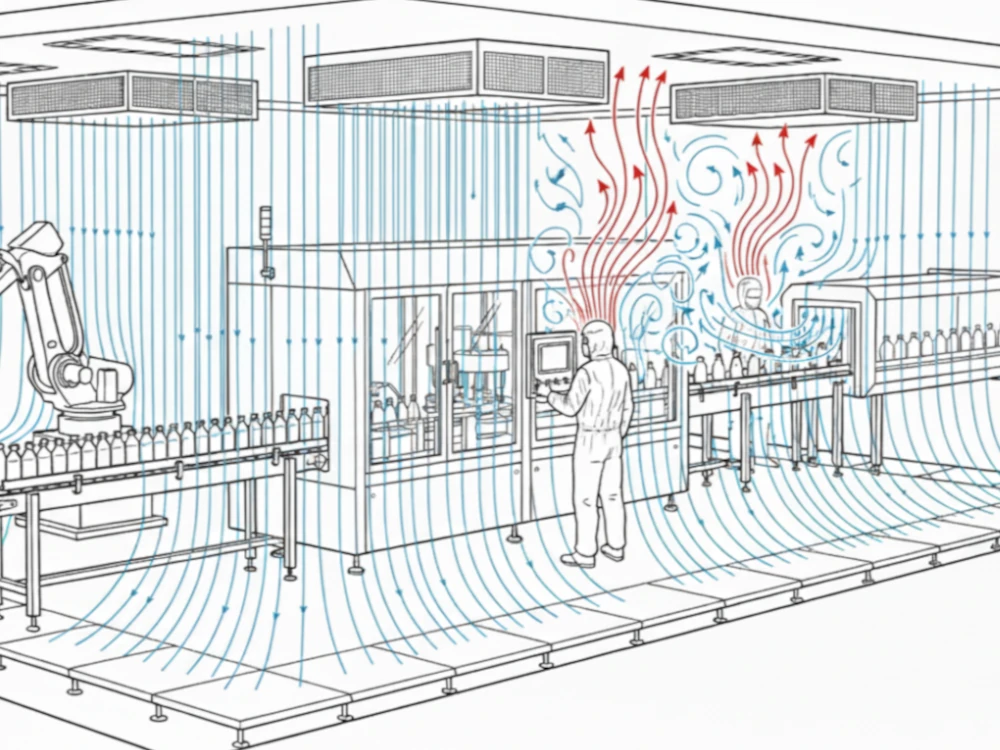
As we scale up single-use technologies (SUT) in 2026, understanding the origins and particularities of these materials is a compliance and sustainability imperative.