The "Green" Single-Use Revolution: Bio-based vs. Fossil-Based
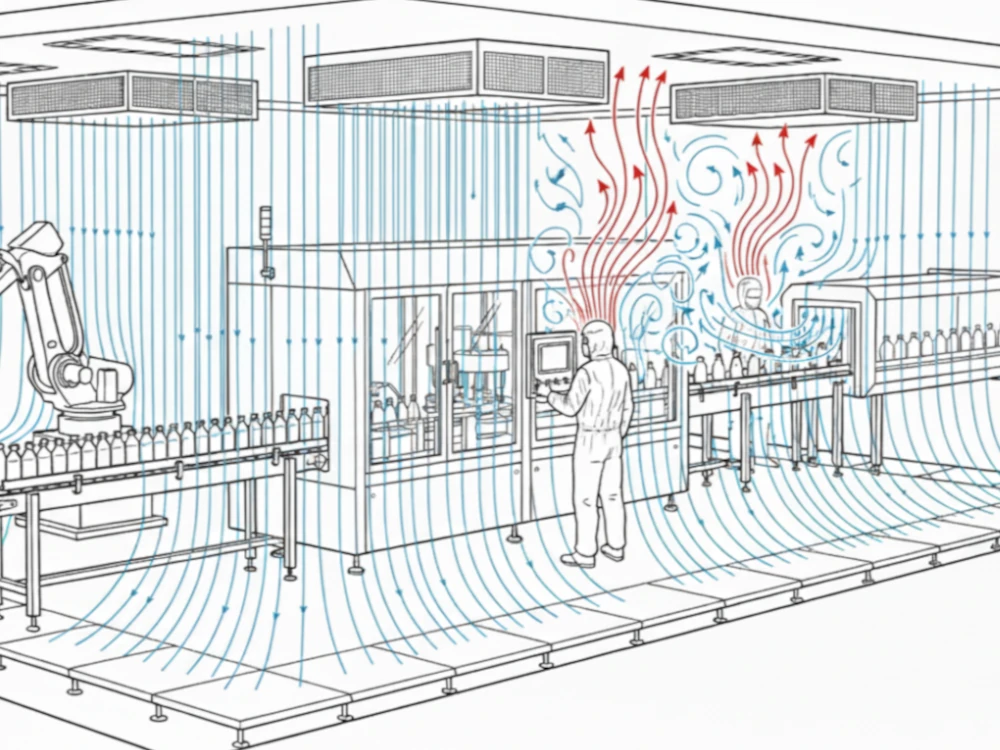
As we move through 2026, the biopharma industry is hitting a major sustainability milestone. For years, the move to Single-Use Technology (SUT) was criticized for its plastic waste. Today, one step towards solving that paradox that has been presented is Bio-based Polymers.